Cancer is a leading cause of death globally. As a result of a growing and aging population, the International Agency for Research on Cancer (IARC), part of the World Health Organization, previously reported that the worldwide cancer burden has risen to 18.1 million new cases and 9.6 million cancer deaths in 2018.
Radiation therapy is commonly used in cancer treatment either alone or combined with surgery or chemotherapy, to kill cancer cells and shrink the tumors. Targeting cancer cells, however, also affects the healthy tissues that surround the tumor, bringing the risk of side effects, some of which may be fatigue, hair loss, skin changes, anemia, nausea and vomiting, depending on the part of the body that is treated.
The development of intensity modulated radiotherapy (IMRT) by linear accelerators (LINACs) allowed the best possible risk/benefit ratio. The LINAC accurately produces, monitors, controls, and conforms the beam of radiation to the planned target and minimizes the effect on normal tissue. IMRT facilitates a range of different doses of radiation across the tumor, achieving the highest intensity possible on tumor cells and lower dose on healthy cells surrounding the area.
The effectiveness of the treatment and the challenges met using LINACs are dependent on:
- The type of cancer
- The size of the tumor
- The location of the tumor
- Radiosensitivity of the tumor and the normal tissue
Conventional linear accelerators are unable to image the tumor and its surrounding healthy tissues when the beam of radiation is applied at the time of treatment. The result is uncertainty about the actual position, and the possibility of movement, of the tumor at the time of treatment. This has led to the use of relatively low doses of radiation at each treatment session so that the total dose of radiation is spread over many treatment sessions, which typically require the patient to visit the hospital on 30 or more occasions over a period of 6 to 10 weeks.
What would happen, though, if we could see what we treat in real time? Magnetic Resonance Imaging-Guided Radiation Therapy (MRIgRT) does this and is now a powerful option in cancer treatment.
In particular, recent advances in radiotherapy utilize a new-generation of hybrid MRI-linear accelerators that facilitate a more widespread adoption of accelerated hypo-fractionated IMRT treatment techniques that are ideally suited not only to the type of restrictions imposed by Covid-19, i.e. the need for more effective treatments requiring fewer hospital visits to reduce the risk of infection of this special category of vulnerable patients, but also to the future of radiotherapy itself, which has now a uniquely powerful and cost-effective tool in the fight against cancer.
Two technologies combined into one device: an MRI scanner and a linear accelerator, together forming the MR-LINAC. Both systems were used separately in radiotherapy for years but now their combination is affecting a revolution in radiation therapy that is likely to change completely the way patients are treated.
The MR-LINAC provides high-quality images of the tumor in real-time which allows us to target and control the radiation dose much more accurately. The device can identify the exact location of the tumor during treatment and adapt the radiation beam according to the movement of the tumor, for example while the patient is breathing. For the treatment of lung or thoracic tumors, the radiation stops automatically during inhalation when the tumor moves and turns back on during exhalation as the tumor returns to its original position. Even when a tumor’s motion is influenced by factors other than the breathing cycle, the system automatically gates (triggers) the beam to stop and start so the beam always coincides only with the tumor. This technique reduces the exposure of health tissue to radiation and delivers the maximum dose precisely to the targeted area, minimizing toxicity.
MRI linear accelerators for the first time allow radiation oncologists to clearly see the tumor, surrounding organs and internal anatomy continuously during the whole treatment, before and while the beam is being applied, providing them with the confidence to more tightly confine the beam of radiation to coincide exactly with the tumor, even while the tumor moves, thus permitting an increase in the dose of radiation to the tumor while at the same time protecting the neighboring healthy tissues, and in fewer treatment sessions than were previously necessary. Instead of the 30 or more treatment sessions previously necessary, typically 5 treatment sessions are sufficient using the technique termed hypo-fractionated MR-Guided Stereotactic Body Radiotherapy (MR-Guided SBRT).
The 1st MR-LINAC ever used worldwide is the MRIdian, manufactured by ViewRay Inc., receiving the CE mark in September 2016, with the 1st patient ever treated in Washington in 2014 and the 2nd is the Unity MR-LINAC by Elekta, clinically implemented in 2017 at University Medical Center (UMC) Utrecht and receiving the CE mark in June 2018. After six years of clinical use, the MRIdian system has treated more than 8,000 patients and performed more than 12,500 adaptive treatments for around 50 ICD-10 cancer codes. Recent social media notifications, articles and publications from hospitals using the MRIdian system during the Covid-19 crisis show the usefulness of hypo-fractioned MR-guided SBRT treatments in reducing the number of necessary hospital visits for cancer patients, as well as some remarkable improvements in the speed and quality of treatment for many types of cancer, for example:

Although Elekta with the Unity system and ViewRay with the MRIdian system are the only two companies at present with certified and commercially available MRI linear accelerators, there have been several developments of MRIgRT devices throughout the years, each with different designs and characteristics, listed in Table 1 below:

The MRIdian system is comprised of three major components: the MRI system, the radiation delivery system, and an integrated treatment planning and delivery software including fully automated beam-gating. It combines 6MV radiation with a 0.35T MRI and uses a split superconductor which allows radiation to pass unimpeded through air to the patient. The Unity system uses a higher field 1.5T MRI scanner and a closed superconductor, which means that the beam travels through metal before reaching the patient. Competition in the market has already started showing positive results, as patients treated with devices by both manufacturers experienced prolongation of survival and reduced radiotoxicity.
At the end of 2019, the 2 companies (Viewray and Elekta) signed a Memorandum of Understanding, with the aim to expand their knowledge in the MRIgRT techniques and to achieve the best personalized clinical results in radiation treatment. The 2 companies will continue to work independently in promoting their designs, but they will also collaborate in clinical trials to get a better understanding on this technological innovation and on how to make it perfectly functional with the minimum operational costs.
According to ViewRay’s financial reports at 31st March 2020, 34 MRIdian devices (4 MRIdian with Cobalt-60 systems and 32 MRIdian with Linac systems) are being used in treatment centers at 35 cancer centers in 13 countries. In addition, six MRIdian Linacs have been delivered to customers that are in varying stages of installation
Sixteen (16) Elekta Unity systems are operated in 11 countries worldwide (Figure1). Forty four percent (44%) of all commercial MR-RTs are located in Europe, followed by 40% in America (19 MR-RTs in the USA and 1 in Canada) and only 10% in Asia, 4% in Middle East and 2% in Australia:
Figure 1. Treatment centers using MRI-linear accelerators around the world.
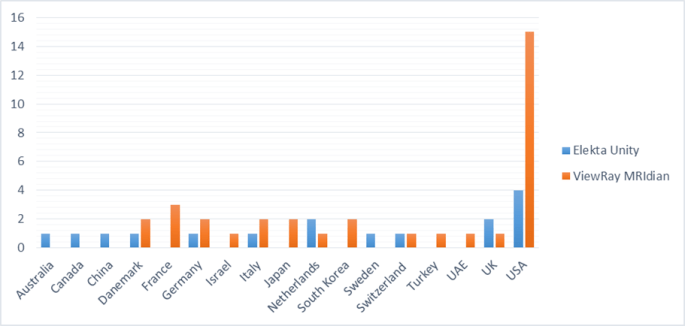
Unfortunately, not all countries have access to the new MR-RT technology yet. As shown in Figure 1, none of the 2 commercially available systems have arrived in Greece, where conventional linear accelerators are being used in radiotherapy.
Despite the advantages of MR-RT in cancer treatment, the technology is still being developed to ensure maximum effectiveness, accuracy and to minimize operational costs and there are several clinical challenges to overcome, some of which are listed below:
- Health economics – The equipment’s elevated costs make it difficult to distribute to all treatment centers and thus limit the access of patients to MRI guided radiotherapy. The actual treatment is more expensive than conventional radiotherapy so patients not requiring access to MRIgRT will be treated using other technological features with high quality results. The cost of MRIgRT will make it difficult for MR-LINACs to become standard-of-care modalities for the foreseeable future.
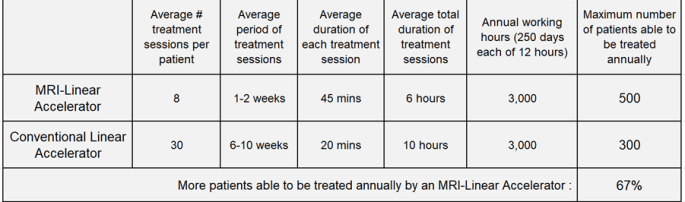
- Treatment time – Although each treatment session is longer in duration on an MRI-linear accelerator fewer treatment sessions in total are required so an MRI-linear accelerator can treat many more patients on an annual basis than can a conventional linear accelerator:
- Practical treatment difficulties – Groups of patients with metal implants or claustrophobia will be impossible to treat under the MRI scanner.
The reduced need for costly travel and accommodation from fewer hospital visits for cancer patients, together with the remarkable improvements in clinical effectiveness now being published from many centers worldwide, reveal the advantages of MRI-linear accelerators for a new type of entirely non-invasive cancer treatment that addresses, effectively, efficiently and at high patient treatment volumes, a wide range of cancers including those, such as pancreatic cancer, that were previously considered to be intractable.
MR guided radiation therapy is already having a dramatic impact on clinical outcomes in cancer treatment and it brings us a step closer to personalized radiation therapy medicine. Some clinical challenges of MR-RT implementation still remain unresolved, but the realization is emerging of the potential for major qualitative and quantitative improvements in the effectiveness and efficiency of cancer treatments from this new paradigm in radiotherapy which may reshape the future of cancer care.
The Greek medical devices and supplies market is slowly recovering
MRI Market in Greece Report 2017
Πρoστατευμένο: Mammography Guide Greece 2017
Ultrasound Systems Contracts in Public Hospitals
Greek Public Hospital Medical Imaging Services Survey Report 2018
